 A recent study published in the British Medical Journal found that results of oncology clinical trials published in major medical journals were described using vague terms that downplayed harms associated with cancer drugs, which was at odds with side effects observed in the actual data. Journalists also use these “vague terms” that minimize potential harms, while emphasizing and exaggerating the potential benefits of new health care interventions. (healthnewsreview.org)
A recent study published in the British Medical Journal found that results of oncology clinical trials published in major medical journals were described using vague terms that downplayed harms associated with cancer drugs, which was at odds with side effects observed in the actual data. Journalists also use these “vague terms” that minimize potential harms, while emphasizing and exaggerating the potential benefits of new health care interventions. (healthnewsreview.org)
Cancer is changing how we test medicines
 1. In a typical drug trial, the manufacturer sends identical pills to patients, to be swallowed. In personalized therapies, patients’ blood samples may be collected, then re-engineered to fight cancer and sent back to the patients. 2. Clinical trials typically start recruiting patients at age 18, but there is a movement to bring young patients into trials. 3. As we pursue cures for rare cancers, studies are getting smaller… (POLITICO)
1. In a typical drug trial, the manufacturer sends identical pills to patients, to be swallowed. In personalized therapies, patients’ blood samples may be collected, then re-engineered to fight cancer and sent back to the patients. 2. Clinical trials typically start recruiting patients at age 18, but there is a movement to bring young patients into trials. 3. As we pursue cures for rare cancers, studies are getting smaller… (POLITICO)
Adolescents, young adults excluded from oncology trials
 In Europe, there are barriers to the inclusion of 12- to 25-year-olds in both adult and pediatric early phase clinical trials. A recent study in France found that only 15% of early phase trials for cancer included teenagers between 12 and 17 years of age. The legal minimum age to participate in adult clinical trials in Europe is 18 years; the authors suggest that such “arbitrary age limits” be eliminated. (MedicalXPress)
In Europe, there are barriers to the inclusion of 12- to 25-year-olds in both adult and pediatric early phase clinical trials. A recent study in France found that only 15% of early phase trials for cancer included teenagers between 12 and 17 years of age. The legal minimum age to participate in adult clinical trials in Europe is 18 years; the authors suggest that such “arbitrary age limits” be eliminated. (MedicalXPress)
Only one-third of drugs in cancer trials meet criteria for “substantial clinical benefit”
 In a recent study, only one-third of the cancer trials that were the basis of FDA approvals in 2016 were found to demonstrate a “substantial clinical benefit” according to the European Society of Medical Oncology Magnitude of Clinical Benefit Scale (ESMO-MCBS). Under the terms of this scale, for a drug to be considered to have a substantial clinical benefit, it not only has to be efficacious, but also has to include information on patient quality of life and postmarketing data. (Cancer Therapy Advisor)
In a recent study, only one-third of the cancer trials that were the basis of FDA approvals in 2016 were found to demonstrate a “substantial clinical benefit” according to the European Society of Medical Oncology Magnitude of Clinical Benefit Scale (ESMO-MCBS). Under the terms of this scale, for a drug to be considered to have a substantial clinical benefit, it not only has to be efficacious, but also has to include information on patient quality of life and postmarketing data. (Cancer Therapy Advisor)
“Bottleneck” of immunotherapy combination trials: 1,000 trials needing 52,000 patients
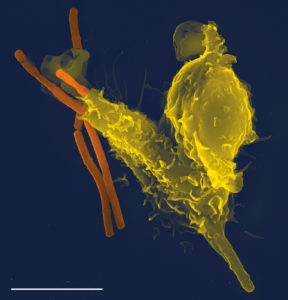 Photo: A scanning electron microscope image of a single neutrophil (yellow), engulfing anthrax bacteria (orange) (Wikipedia).
Photo: A scanning electron microscope image of a single neutrophil (yellow), engulfing anthrax bacteria (orange) (Wikipedia).
A bottleneck of immunotherapy clinical trials has put the field’s advancement on hold. Over 1,000 clinical trials are lined up to test PD-1- or PD-L1-based combinations, projecting to enroll a total of 52,000 patients. According to Tom Schuetz, CEO of Compass Therapeutics, it’s impossible to enroll those trials, and we need to do a better job about evaluating these combinations preclinically. “We fundamentally believe in just doing a ton of preclinical experiments.” (Fierce Biotech)
NCI funding shortchanges gynecological cancer research
 According to a presentation at the Society of Gynecologic Oncology, federal funding for medical research shortchanges gynecologic oncology when cancer lethality is taken into account. Among 13 common cancers included in the analysis, gynecologic cancers ranked near the bottom in National Cancer Institute (NCI) funding support, as measured by spending per years of life lost (YLL).
According to a presentation at the Society of Gynecologic Oncology, federal funding for medical research shortchanges gynecologic oncology when cancer lethality is taken into account. Among 13 common cancers included in the analysis, gynecologic cancers ranked near the bottom in National Cancer Institute (NCI) funding support, as measured by spending per years of life lost (YLL).
HIV patients w/cancer less likely to receive appropriate care
![]() Results from a survey of U.S. oncologists suggest that patients with cancer and HIV are not likely to receive optimal cancer care that takes into consideration their HIV status. Seventy percent of oncologists said that sufficient guidelines for treating these patients were not available; part of the problem is that patients with HIV are often excluded from cancer clinical trials, leading to a gap in knowledge about these patients.
Results from a survey of U.S. oncologists suggest that patients with cancer and HIV are not likely to receive optimal cancer care that takes into consideration their HIV status. Seventy percent of oncologists said that sufficient guidelines for treating these patients were not available; part of the problem is that patients with HIV are often excluded from cancer clinical trials, leading to a gap in knowledge about these patients.
Most new clinical trials are in oncology, Immune-oncology drugs predominate
![]() According to Global Data, oncology is the top therapy area for clinical trials initiated in 2018; most industry sponsored trials are for solid tumors; and, among all clinical trials, immune oncology drugs are the most common. North America, Europe and Asia-Pacific account for more than 90% of all trial locations, with most of the North American trial sites based in the U.S. (CLINICAL LEADER)
According to Global Data, oncology is the top therapy area for clinical trials initiated in 2018; most industry sponsored trials are for solid tumors; and, among all clinical trials, immune oncology drugs are the most common. North America, Europe and Asia-Pacific account for more than 90% of all trial locations, with most of the North American trial sites based in the U.S. (CLINICAL LEADER)
Phase 1 pediatric cancer trials are therapeutic. True? Or, is this “a noble lie”?
 It’s this author’s view that it may be a mistake to think of early phase trials as having a therapeutic impetus. U.S. regulations require that, when we enroll children, we view phase 1 trial participation as therapeutic. However, a recent meta-analysis concluded that phase 1 trials in children cannot generally be presented as having a therapeutic risk/benefit. Thus, phase 1 trials in children cannot be reconciled with research regulations. (translationalethics.com)
It’s this author’s view that it may be a mistake to think of early phase trials as having a therapeutic impetus. U.S. regulations require that, when we enroll children, we view phase 1 trial participation as therapeutic. However, a recent meta-analysis concluded that phase 1 trials in children cannot generally be presented as having a therapeutic risk/benefit. Thus, phase 1 trials in children cannot be reconciled with research regulations. (translationalethics.com)
More patients are joining Phase 1 cancer trials – to get access to new experimental treatments
![]() The importance of early stage clinical trials in oncology is increasing. Historically, cancer patients participated in phase 1 trials because this was the only option for those with advanced cancer that would not respond to standard treatment. Today, more patients are interested in participating not because it is a last resort for them, but because they gain access to new treatment alternatives to chemotherapy, and there are larger trials which benefit more patients. (SCIENMAG)
The importance of early stage clinical trials in oncology is increasing. Historically, cancer patients participated in phase 1 trials because this was the only option for those with advanced cancer that would not respond to standard treatment. Today, more patients are interested in participating not because it is a last resort for them, but because they gain access to new treatment alternatives to chemotherapy, and there are larger trials which benefit more patients. (SCIENMAG)
