 A study published in JAMA Oncology found that 1 in 6 cancer drugs approved between 2013 and 2018 were supported by randomized controlled trials (RCTs) that do not prove the drug’s superiority over the standard of care, as their control arms did not accurately represent the standard of care. “The choice of control arm should be optimized to ensure that new cancer agents are truly superior to what most clinicians would prescribe outside a clinical trial setting. (docwire)
A study published in JAMA Oncology found that 1 in 6 cancer drugs approved between 2013 and 2018 were supported by randomized controlled trials (RCTs) that do not prove the drug’s superiority over the standard of care, as their control arms did not accurately represent the standard of care. “The choice of control arm should be optimized to ensure that new cancer agents are truly superior to what most clinicians would prescribe outside a clinical trial setting. (docwire)
Use of surrogate end points shortens cancer trials
The use of surrogate end points (progression-free survival) (PFS) in oncology trials reduced the time needed to conduct clinical trials by 11 months compared to the study duration required to assess the overall survival (OS) benefit of a cancer drug. (MedicalResearch.com)
Protocol amendments may be unnecessary
According to an expert, the rise in the number of protocol amendments occurring before the first patient has even received the first dose reflects a failure in planning trial design. Amending a protocol often involves making the study more complex, which impacts study costs and participant satisfaction. (CenterWatch)
Multiple expansion cohort clinical trials: a new study design
www.appliedclinicaltrialsonline.com
The rapid rise of targeted drugs and biologics in oncology has led to the creation of a variation to the typical trial design that can include multiple expansion cohorts. A multiple expansion cohort trial allows sponsors who spot positive responses to an investigational drug during early stages of testing to expand the trial using additional single-arm studies; these designs can cut the typical development times for conventional trials in half. (Applied Clinical Trials)
More data will be generated in 2019 than in the last 5,000 years
The new year will bring more data than the last 500 decades, information that will be used to improve clinical trial design and shorten drug development timelines, say industry experts. (outsourcing-pharma.com)
Blogger with cystic fibrosis (CF) excluded from CF clinical trials
 Ella Balasa blog. Ms. Balasa is ineligible for clinical trials for treatment of symptoms of cystic fibrosis (CF) because most CF studies require participants to have a forced expiratory volume in one second (FEV1) of at least 40 percent, and hers is 25 percent. She suggests that FEV1 is an insufficient proxy for identifying “healthy” candidates for CF trials, and that current inclusion and exclusion practices be adjusted. (STAT)
Ella Balasa blog. Ms. Balasa is ineligible for clinical trials for treatment of symptoms of cystic fibrosis (CF) because most CF studies require participants to have a forced expiratory volume in one second (FEV1) of at least 40 percent, and hers is 25 percent. She suggests that FEV1 is an insufficient proxy for identifying “healthy” candidates for CF trials, and that current inclusion and exclusion practices be adjusted. (STAT)
Cancer is changing how we test medicines
 1. In a typical drug trial, the manufacturer sends identical pills to patients, to be swallowed. In personalized therapies, patients’ blood samples may be collected, then re-engineered to fight cancer and sent back to the patients. 2. Clinical trials typically start recruiting patients at age 18, but there is a movement to bring young patients into trials. 3. As we pursue cures for rare cancers, studies are getting smaller… (POLITICO)
1. In a typical drug trial, the manufacturer sends identical pills to patients, to be swallowed. In personalized therapies, patients’ blood samples may be collected, then re-engineered to fight cancer and sent back to the patients. 2. Clinical trials typically start recruiting patients at age 18, but there is a movement to bring young patients into trials. 3. As we pursue cures for rare cancers, studies are getting smaller… (POLITICO)
FDA wants sponsors to use patient-reported outcomes in phase 3 trials
 Patient-reported outcomes (PROs) can enhance drug development, but the integration of PRO measures as end points in phase 3 clinical trials has been slow. The FDA held a two-day workshop to gather input on a series of drug development documents they are developing to provide guidance to stakeholders on collecting information on patient experiences to inform product development. (ajmc.com)
Patient-reported outcomes (PROs) can enhance drug development, but the integration of PRO measures as end points in phase 3 clinical trials has been slow. The FDA held a two-day workshop to gather input on a series of drug development documents they are developing to provide guidance to stakeholders on collecting information on patient experiences to inform product development. (ajmc.com)
Should digital health startups be required to do randomized controlled trials?
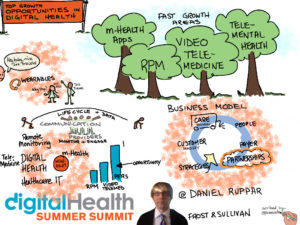 Health care startups are attracting billions of dollars in risk capital, but most do not pursue the validation of a randomized clinical trial. Reflexion, a digital heal care startup, conducted a prospective randomized controlled trial of their product. They concluded that this method is not suited for the typical digital health startup, and propose that we streamline and accelerate the process for collecting proof of value for digital health startups. (statnews.com)
Health care startups are attracting billions of dollars in risk capital, but most do not pursue the validation of a randomized clinical trial. Reflexion, a digital heal care startup, conducted a prospective randomized controlled trial of their product. They concluded that this method is not suited for the typical digital health startup, and propose that we streamline and accelerate the process for collecting proof of value for digital health startups. (statnews.com)
ISCHEMIA trial raises controversy over the use of composite endpoints in clinical trials
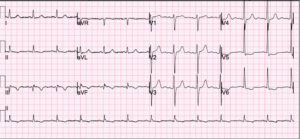 A special session at a recent TCT (Transcatheter Cardiovascular Therapeutics) meeting addressed the question of how to design clinical trials in interventional cardiovascular medicine in an era where new therapies offer incremental gains. The pitfalls of using composite endpoints in studies were discussed in the context of the ISCHEMIA trial, which added additional “soft” endpoints late in the design phase. The program session also discussed the feasibility of sham-controlled trials for medical devices. (tctMD)
A special session at a recent TCT (Transcatheter Cardiovascular Therapeutics) meeting addressed the question of how to design clinical trials in interventional cardiovascular medicine in an era where new therapies offer incremental gains. The pitfalls of using composite endpoints in studies were discussed in the context of the ISCHEMIA trial, which added additional “soft” endpoints late in the design phase. The program session also discussed the feasibility of sham-controlled trials for medical devices. (tctMD)
