 According to a recent study, if the eligibility criteria of ALS targeted clinical trials were applied to a population of ALS patients in the Netherlands, nearly 60% of these patients would have been excluded from clinical studies. The authors recommend that we use a prediction model to select patients for clinical trials, rather than standard eligibility approaches. (ALS NEWS TODAY)
According to a recent study, if the eligibility criteria of ALS targeted clinical trials were applied to a population of ALS patients in the Netherlands, nearly 60% of these patients would have been excluded from clinical studies. The authors recommend that we use a prediction model to select patients for clinical trials, rather than standard eligibility approaches. (ALS NEWS TODAY)
Trials exclude patients with mental or psychiatric disorders
 According to the FDA, nearly 50% of investigational new drug applications excluded patients with mental or psychiatric disorders. Should cancer clinical trial eligibility regarding mental illness be relaxed? One viewpoint is that we should relax enrollment criteria and avoid unnecessary exclusion of patients with serious mental illness. (Healio)
According to the FDA, nearly 50% of investigational new drug applications excluded patients with mental or psychiatric disorders. Should cancer clinical trial eligibility regarding mental illness be relaxed? One viewpoint is that we should relax enrollment criteria and avoid unnecessary exclusion of patients with serious mental illness. (Healio)
National Cancer Institute expands eligibility criteria for trials
 The National Cancer Institute (NCI) has revised its clinical trial protocols to expand access for previously excluded patients. The revisions expand access for potential participants with preexisting conditions in 4 categories, and patients under the age of 18 years. (OncLive)
The National Cancer Institute (NCI) has revised its clinical trial protocols to expand access for previously excluded patients. The revisions expand access for potential participants with preexisting conditions in 4 categories, and patients under the age of 18 years. (OncLive)
ASCO policy statement addresses financial barriers to clinical trial participation
 The American Society for Clinical Oncology (ASCO) has released a new policy statement, “Addressing Financial Barriers to Patient Participation in Clinical Trials”, which makes several recommendations to address financial barriers to trial participation. Among other recommendations, the statement recommends improving payer clinical trial coverage, and transparency regarding out-of-pocket costs. (Healio)
The American Society for Clinical Oncology (ASCO) has released a new policy statement, “Addressing Financial Barriers to Patient Participation in Clinical Trials”, which makes several recommendations to address financial barriers to trial participation. Among other recommendations, the statement recommends improving payer clinical trial coverage, and transparency regarding out-of-pocket costs. (Healio)
ASCO, FDA recommend relaxing cancer trial eligibility criteria
 A new study found that physicians were less likely to offer clinical trial participation to patients with a comorbidity. The American Society of Clinical Oncology (ASCO), Friends of Cancer Research, and the FDA recently reviewed the comorbid conditions that are generally used in eligibility criteria to exclude patients, and recommended that many be modified or liberalized, which could be necessary to improve study participation. (MEDPAGE TODAY)
A new study found that physicians were less likely to offer clinical trial participation to patients with a comorbidity. The American Society of Clinical Oncology (ASCO), Friends of Cancer Research, and the FDA recently reviewed the comorbid conditions that are generally used in eligibility criteria to exclude patients, and recommended that many be modified or liberalized, which could be necessary to improve study participation. (MEDPAGE TODAY)
Clinical research industry’s 3 wishes for 2019
What might the clinical research industry wish for in 2019 to improve their ability to bring new products to market? Innovation in patient recruitment; blockchain to improve clinical research data storage; and a greater variety of eClinical platforms to meet the needs of researchers in different areas. (medrio)
Blogger with cystic fibrosis (CF) excluded from CF clinical trials
 Ella Balasa blog. Ms. Balasa is ineligible for clinical trials for treatment of symptoms of cystic fibrosis (CF) because most CF studies require participants to have a forced expiratory volume in one second (FEV1) of at least 40 percent, and hers is 25 percent. She suggests that FEV1 is an insufficient proxy for identifying “healthy” candidates for CF trials, and that current inclusion and exclusion practices be adjusted. (STAT)
Ella Balasa blog. Ms. Balasa is ineligible for clinical trials for treatment of symptoms of cystic fibrosis (CF) because most CF studies require participants to have a forced expiratory volume in one second (FEV1) of at least 40 percent, and hers is 25 percent. She suggests that FEV1 is an insufficient proxy for identifying “healthy” candidates for CF trials, and that current inclusion and exclusion practices be adjusted. (STAT)
New machine learning tool could reduce clinical trial attrition
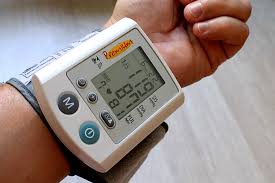 Cambridge Consultants has designed a digital health tool that aims to help prevent patients becoming stressed and dropping out of clinical trials. The tool, known as Verum, uses biometric data and machine learning to measure and monitor a participant’s stress levels during trials, enabling trial coordinators to intervene and mitigate the effect of stress on compliance. (pharmaphorum)
Cambridge Consultants has designed a digital health tool that aims to help prevent patients becoming stressed and dropping out of clinical trials. The tool, known as Verum, uses biometric data and machine learning to measure and monitor a participant’s stress levels during trials, enabling trial coordinators to intervene and mitigate the effect of stress on compliance. (pharmaphorum)
Hospitals band together to attract drug trials

City hospital building with ambulance and helicopter in flat design.
Regional hospitals across the U.S. have formed consortiums to allow them to compete with urban medical centers for clinical trials and boost their clinical trial revenue. For pharma companies, centralizing the process of drug testing across several hospitals can provide them with increased access to trial participants, and requires fewer institutional review board (IRB) reviews. (Bloomberg)
Lack of communication in clinical trial teams
www.journalofclinicalpathways.com
Less than 2% of adult cancer patients are enrolling in clinical trials. What are the barriers contributing to this crisis that are related to the science of clinical trials? One barrier is “clinical trial procedures and conduct”- the team interactions between clinical and research teams suffered from lack of communication, team rivalries, and other inefficiencies. (J of Clinical Pathways)
